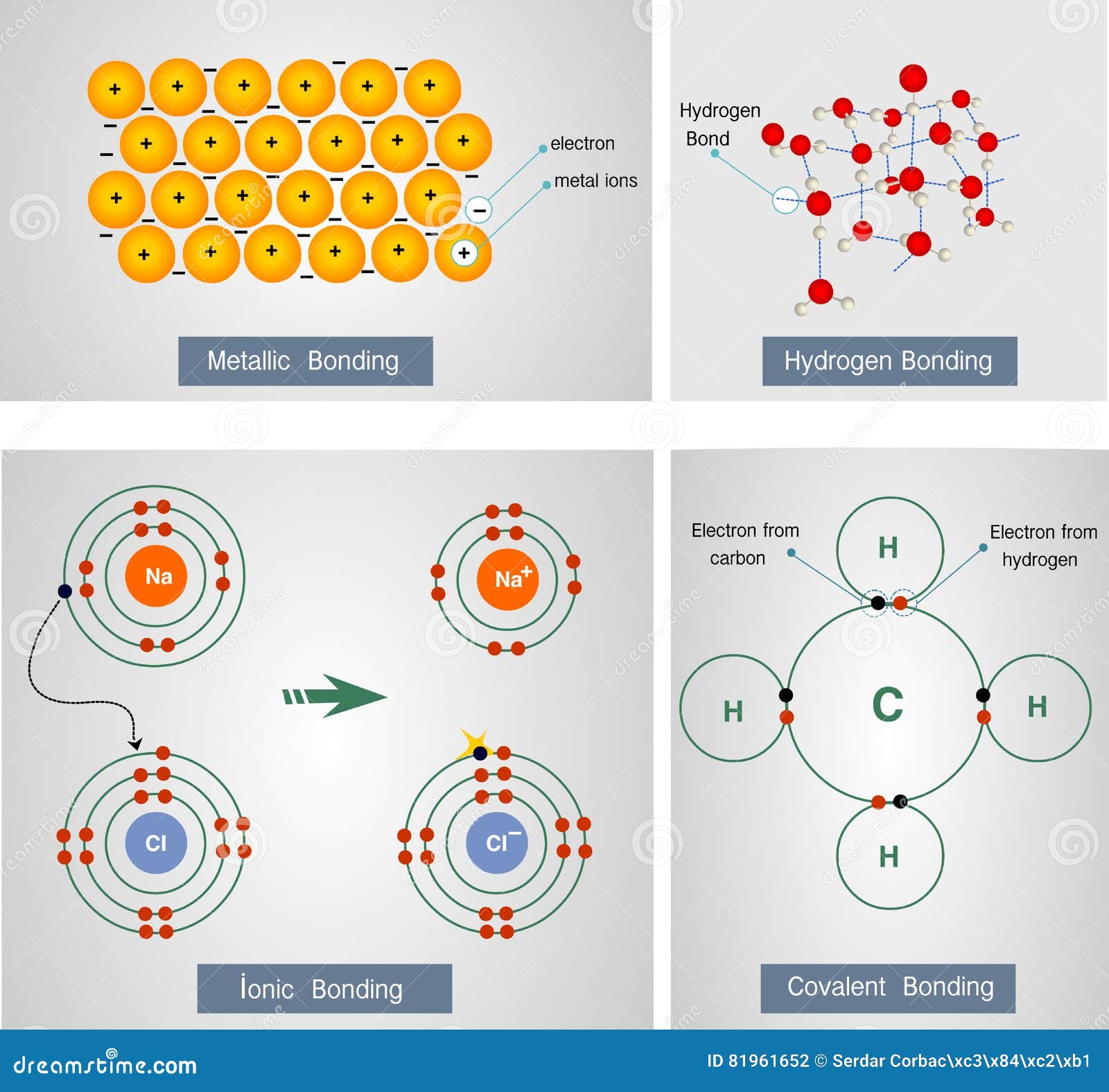 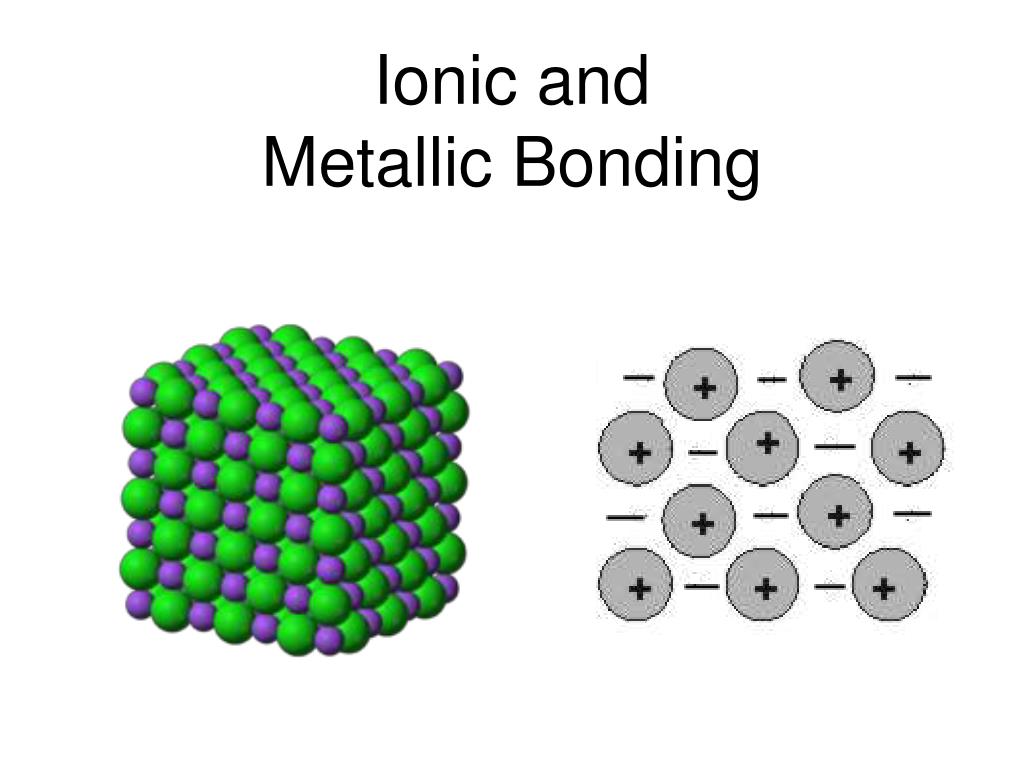
Many people use the term “sea of electrons” to refer to this shared pool of valence electrons. All the atoms in a piece of metal share these electrons, and they can flow freely through the whole material. As a result, when there are only metal atoms around, the valence electrons of one atom are hard to distinguish from those of its neighbors. This is the reason why they give up valence electrons around electronegative atoms to form ionic compounds. Metallic elements have low electronegativity, which means that they hold onto their valence electrons loosely. In substances containing only metal atoms, however, the interaction involved has a different character, and we call it metallic bonding. Bonding between metals and nonmetals is usually ionic, while bonding among nonmetals is usually covalent. But what are the characteristics of this kind of bonding? First, as the name implies, this type of bonding is found in metals and metal alloys. Metallic bonds are not discrete directional bonds between specific atoms, so it often makes sense to talk about metallic “bonding” rather than individual bonds. In the rest of this article, we will take a look at the different parts of this definition and break down what it means, and explore the consequences of metallic bonding interactions! What is a Metallic Bond? The short answer: metallic bonding is a type of chemical bonding between two or more metal atoms, which arises from the attraction between positively charged metal nuclei and their delocalized valence electrons. In this tutorial, you will learn about metallic bonding, its characteristics and properties, and even learn some examples! Topics Covered in Other Articles
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
